Enthalpy vs entropy11/23/2023  
The process can be exothermic, where heat (q) is negative and is released by the system into the surrounding. Heat (q) (at constant pressure) = Enthalpy (H)Īny of the terms in the above equations may either be positive or negative. Heat (q) (at constant volume) = Internal energy (U) 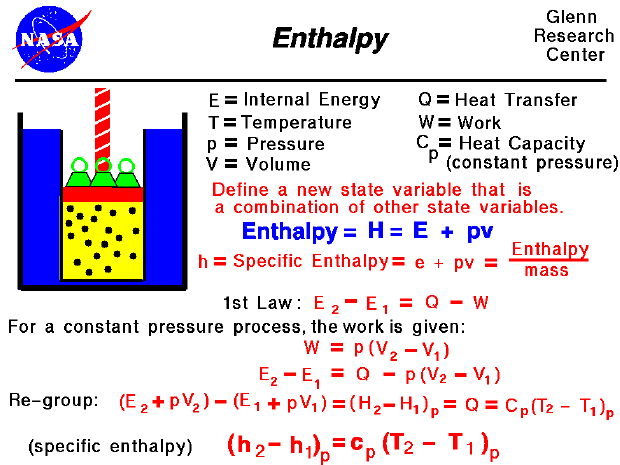
If q is the amount of heat absorbed by the system from surroundings: Concepts Berg Internal energy vs Enthalpy on operational basis.Internal energy vs Enthalpy on operational basis.H can be applied to refrigerators (freezing), boilers (boiling) etc. A glass of water has no apparent energy but it does have internal energy. 
It is an energy change between the system and the surroundings. Internal energy is the total energy in a system. Enthalpy of an ideal gas is 40% more than internal energy. The internal energy of the ideal gas is 40% less than enthalpy. U=0 At constant temperature, H=0 because U=0 and pressure-volume remain constant. Enthalpy is a function of temperature and pressure. Internal energy is a function of temperature. It is an energy change between two states. According to the formula, enthalpy isĮqual to the sum of internal energy and product of pressure-volume work. H=U+PV According to the formula, change in internal energy is equal to heat transfer to the system minus work done. For open systems, the term enthalpy is used. Negative H For closed systems, the term internal energy is used. Systems absorb energy and decreases when systems release energy to the surrounding. Internal Energy Enthalpy It is the sum of kinetic energyĪnd potential energy. 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
